In the medical device industry, cable integrity is not a matter of convenience — it is a matter of patient safety. From surgical robots and MRI machines to portable patient monitors and infusion pumps, every cable that carries power or data must perform flawlessly under repeated sterilization cycles, mechanical stress, and strict biocompatibility requirements. Medical device cable protection with heat shrink tubing has become the gold standard solution for OEMs, contract manufacturers, and hospital biomedical engineering teams worldwide.
At Willele Electric, we manufacture precision heat shrink tubes engineered specifically for medical-grade applications, meeting ISO 10993 biocompatibility standards and the demanding mechanical requirements of Class II and Class III medical devices. This guide covers everything procurement engineers, device designers, and biomedical technicians need to know about selecting and applying heat shrink tubing for medical cable protection.
Why Cable Protection Is Critical in Medical Devices
Medical device cables endure abuse that would destroy standard commercial wiring within weeks. Consider the lifecycle of a typical surgical instrument cable: it is coiled and uncoiled hundreds of times per year, subjected to autoclave sterilization at 134°C and 3 bar pressure, exposed to disinfectant chemicals, and flexed repeatedly at connector entry points where stress concentration is highest.
Without adequate protection, three failure modes dominate:
Insulation cracking at flex points — Repeated bending at connector strain relief zones fatigues the cable jacket, creating micro-cracks that allow fluid ingress and eventual short circuits.
Connector pull-out — Mechanical stress at the cable-to-connector interface gradually separates the bond between cable jacket and connector housing, exposing conductors.
Chemical degradation — Disinfectants containing quaternary ammonium compounds, glutaraldehyde, and hydrogen peroxide attack standard PVC and polyethylene insulation, causing swelling, cracking, and loss of dielectric properties.
Heat shrink tubing addresses all three failure modes by adding a robust secondary protective layer precisely where it is needed most.
Medical-Grade Heat Shrink Tubing: Material Selection Guide
Material choice determines biocompatibility, sterilization compatibility, and mechanical performance. The following table compares the most widely used materials in medical device applications:
| Materiale | Rapporto di restringimento | Operating Temp | Sterilization Compatibility | Biocompatibility | Key Advantage | Typical Application |
|---|---|---|---|---|---|---|
| Polyolefin (Standard) | 2:1 / 3:1 | -55°C to 135°C | EtO, Gamma | ISO 10993 compliant | Cost-effective, flexible | General cable bundling |
| Polyolefin (Dual-Wall) | 3:1 | -55°C to 110°C | EtO, Gamma | ISO 10993 compliant | Adhesive inner layer seals moisture | Connector strain relief |
| PTFE | 4:1 | -200°C to 260°C | All methods incl. autoclave | Eccellente | Chemical inertness, autoclave-safe | Surgical instruments, autoclavable devices |
| FEP | 1.6:1 | -200°C to 200°C | All methods incl. autoclave | Eccellente | Transparent, autoclave-safe | Endoscope cables, visualization systems |
| PVDF | 2:1 | -60°C to 175°C | EtO, Gamma, autoclave | ISO 10993 compliant | High abrasion resistance | Robotic surgical arms |
| Silicone | 2:1 | -60°C to 200°C | All methods incl. autoclave | Eccellente | Extreme flexibility | Patient contact cables, wearables |
| PET (Braided) | N/A | -70°C to 150°C | EtO, Gamma | ISO 10993 compliant | Mechanical abrasion protection | High-flex cable harnesses |
Understanding the Cross-Section: How Heat Shrink Tubing Protects Medical Cables
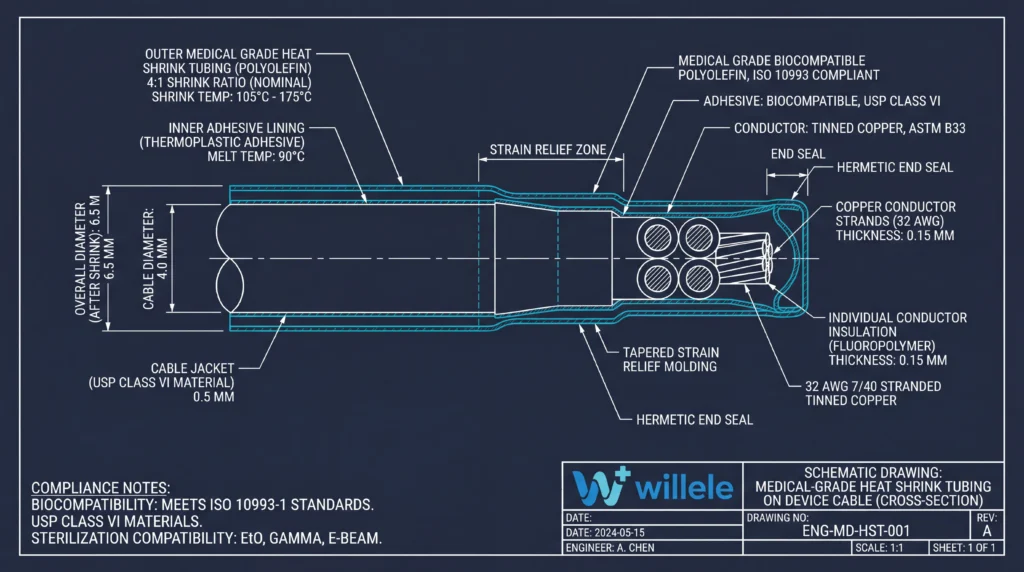
Alt text: Technical engineering cross-section schematic diagram of Willele medical-grade heat shrink tubing showing all internal layers including outer polyolefin shrink layer, adhesive inner lining, cable jacket, individual conductor insulation, copper conductor strands, and labeled strain relief and end seal zones with ISO 10993 compliance annotations
The cross-section of a properly applied dual-wall medical heat shrink assembly reveals a precisely engineered multi-layer protection system:
- Outer shrink layer: Provides abrasion resistance, chemical barrier, and color-coded circuit identification
- Adhesive inner layer: Melts during shrinking to flow into surface irregularities, creating a hermetic moisture seal
- Transition zone: Gradual diameter change distributes flex stress over a longer cable length, preventing stress concentration
- End seals: Adhesive overflow at both ends creates waterproof terminations rated to IP67 or higher
This construction is why dual-wall heat shrink tubing consistently outperforms single-wall alternatives in IEC 60601-1 mechanical stress testing.
Key Performance Standards for Medical Heat Shrink Tubing
Compliance with the following standards is non-negotiable for medical device applications:
| Standard | Scope | Relevance to Heat Shrink Tubing |
|---|---|---|
| ISO 10993-1 | Biological evaluation of medical devices | Biocompatibility testing framework |
| ISO 10993-5 | Cytotoxicity testing | Ensures tubing materials are non-cytotoxic |
| ISO 10993-10 | Sensitization and irritation | Skin-contact cable applications |
| IEC 60601-1 | Medical electrical equipment safety | Mechanical and electrical performance |
| UL 224 | Heat shrink tubing listing | Electrical insulation performance |
| RoHS / REACH | Hazardous substance restriction | Halogen-free and heavy metal-free compliance |
| FDA 21 CFR | Medical device regulation (USA) | Material traceability requirements |
| EU MDR 2017/745 | Medical device regulation (Europe) | Technical documentation requirements |
Willele Electric maintains full material traceability documentation and third-party test reports for all medical-grade heat shrink product lines, supporting FDA 510(k) submissions and EU MDR technical files.
Application Guide: Heat Shrink Tubing for Common Medical Cable Types
Patient Monitoring Cables (ECG, SpO₂, NIBP)
Patient monitoring cables present the highest flex cycle requirements in clinical settings — a typical ECG lead wire undergoes 50,000+ flex cycles annually. Specify 2:1 or 3:1 polyolefin tubing at connector strain relief points with a recovered wall thickness of at least 0.5mm. For cables with patient contact classification (Type CF or BF per IEC 60601-1), select tubing with confirmed ISO 10993-5 cytotoxicity compliance.
Surgical Instrument Cables (Electrosurgical, Ultrasonic)
Electrosurgical and ultrasonic instrument cables must withstand autoclave sterilization at 134°C. Only PTFE or FEP heat shrink tubing provides the necessary thermal stability. PTFE’s 4:1 shrink ratio enables tight coverage over irregular connector geometries common in laparoscopic instruments.
Imaging System Cables (Ultrasound, Endoscope)
Imaging cables contain dozens of fine coaxial conductors requiring precise impedance control. FEP heat shrink tubing is preferred for its transparency (allowing visual inspection of underlying conductors) and low dielectric constant, which minimizes signal degradation in high-frequency ultrasound applications.
Implantable Device Lead Cables
Implantable leads require materials with decades of in-vivo stability. While heat shrink tubing is primarily used during manufacturing and testing rather than in the final implanted assembly, silicone heat shrink tubing provides the biocompatibility and flexibility needed for pre-implant handling protection.
Sterilization Compatibility Comparison
| Sterilization Method | Max Temp | Poliolefina | PTFE | FEP | PVDF | Silicone |
|---|---|---|---|---|---|---|
| Ethylene Oxide (EtO) | 60°C | ✅ Excellent | ✅ Excellent | ✅ Excellent | ✅ Excellent | ✅ Excellent |
| Gamma Irradiation | Ambient | ✅ Good | ✅ Excellent | ✅ Excellent | ✅ Good | ⚠️ Degrades over time |
| Steam Autoclave | 134°C | ❌ Not suitable | ✅ Excellent | ✅ Excellent | ✅ Good | ✅ Excellent |
| Hydrogen Peroxide Plasma | 50°C | ✅ Good | ✅ Excellent | ✅ Excellent | ✅ Good | ✅ Good |
| Chemical Disinfection | Ambient | ⚠️ Material-dependent | ✅ Excellent | ✅ Excellent | ✅ Excellent | ✅ Good |
Installation Best Practices for Medical Applications
Proper application technique is as important as material selection. Incorrect installation voids performance ratings and can introduce new failure points.
Pre-installation preparation: Clean cable surfaces with isopropyl alcohol and allow to dry completely. Any contamination between the cable jacket and heat shrink tubing prevents adhesive bonding and creates moisture ingress pathways.
Tubing size selection: Select tubing with a supplied inner diameter 10–20% larger than the cable outer diameter. Oversized tubing fails to achieve full shrinkage and leaves voids; undersized tubing cannot be installed without stretching, which stresses the material.
Heat application: Use a calibrated heat gun with temperature control, not an open flame. Apply heat evenly by moving the gun continuously along the tubing length. For dual-wall tubing, maintain heat until adhesive flows visibly from both ends — this confirms complete activation of the inner adhesive layer.
Quality verification: After cooling, inspect for complete shrinkage with no bubbles or voids, confirm adhesive flow at both ends, and perform a pull test to verify strain relief engagement.
Willele Medical Heat Shrink Product Line Comparison
| Product Series | Materiale | Rapporto di restringimento | Wall Thickness (Recovered) | Temp Range | ISO 10993 | Il migliore per |
|---|---|---|---|---|---|---|
| WL-MED-PO | Poliolefina | 2:1 | 0.4mm | -55°C~135°C | ✅ | General medical cables |
| WL-MED-DW | Dual-wall polyolefin | 3:1 | 0.6mm | -55°C~110°C | ✅ | Connector strain relief |
| WL-MED-PT | PTFE | 4:1 | 0.3mm | -200°C~260°C | ✅ | Autoclavable instruments |
| WL-MED-FP | FEP (transparent) | 1.6:1 | 0.4mm | -200°C~200°C | ✅ | Imaging/endoscope cables |
| WL-MED-SI | Silicone | 2:1 | 0.8mm | -60°C~200°C | ✅ | Patient contact, wearables |
| WL-MED-PV | PVDF | 2:1 | 0.5mm | -60°C~175°C | ✅ | Robotic surgical systems |
Frequently Asked Questions
Q: Does heat shrink tubing need to be biocompatible for all medical device cable applications?
A: Biocompatibility testing is required whenever tubing contacts the patient directly or indirectly through prolonged skin contact. For internal device wiring with no patient contact, standard UL 224-listed polyolefin tubing is generally acceptable. Always consult your regulatory affairs team to determine the applicable ISO 10993 test matrix for your specific device classification.
Q: Can heat shrink tubing be used on cables that will be autoclaved?
A: Yes, but only with PTFE, FEP, or silicone heat shrink tubing. Standard polyolefin tubing softens and deforms at autoclave temperatures of 121–134°C. Willele’s WL-MED-PT (PTFE) and WL-MED-FP (FEP) series are specifically validated for repeated autoclave cycles without dimensional change or property degradation.
Q: What shrink ratio should I specify for medical connector strain relief?
A: For connector strain relief applications, specify a minimum 3:1 shrink ratio with a dual-wall construction. The higher shrink ratio ensures complete coverage over the transition from connector housing to cable jacket, while the adhesive inner layer creates the hermetic seal necessary to prevent fluid ingress during cleaning and sterilization.
Q: How do I document heat shrink tubing in an FDA 510(k) submission?
A: Your technical file should include the tubing manufacturer’s material datasheet, ISO 10993 biocompatibility test reports, UL 224 listing certificate, and material traceability documentation. Willele Electric provides a complete regulatory documentation package for all medical-grade product lines, including extractables and leachables data where required.
Q: What is the minimum order quantity for custom-colored or custom-printed medical heat shrink tubing?
A: Willele Electric offers custom color matching and logo/text printing on medical heat shrink tubing with minimum order quantities starting from 500 meters per SKU. Custom colors support color-coded circuit identification systems required by IEC 60601-1 for multi-conductor medical cables.



